How TMS Works on the Brain Part 7:Subcortical Effects of TMS on Neural Pathways
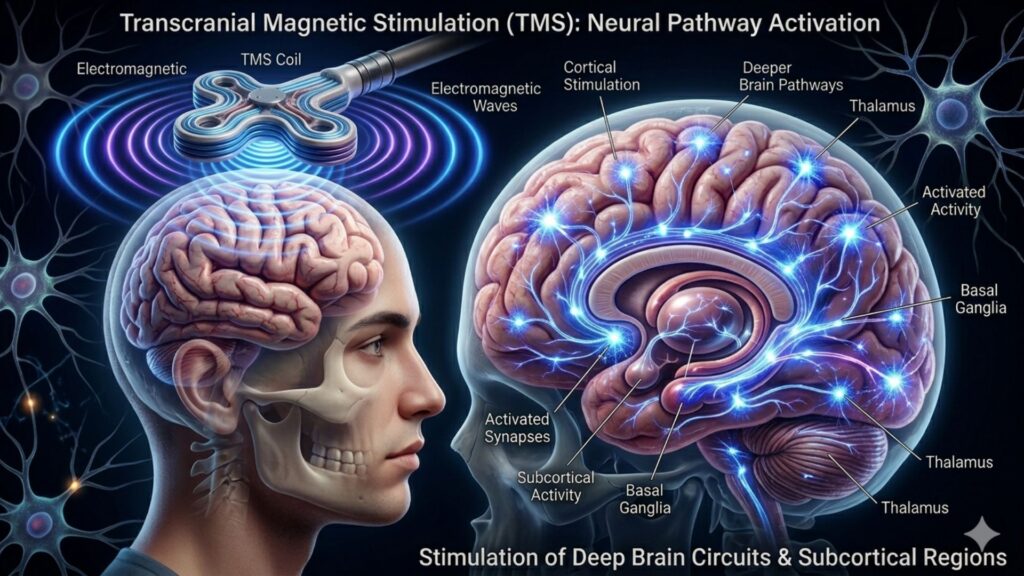
Transcranial magnetic stimulation (TMS) is widely known for its ability to influence the outer layer of the brain—the cerebral cortex. However, research has shown that the effects of TMS extend much deeper, reaching into the brain’s subcortical regions, which include critical structures such as the thalamus, amygdala, hippocampus, and basal ganglia. These areas play vital roles in emotion, memory, reward processing, and motor control, and their involvement helps explain the widespread clinical benefits of TMS.
Because TMS delivers magnetic pulses to the surface of the brain, it might seem counterintuitive that it could affect areas buried deep within. Yet the brain is a highly interconnected organ. When TMS stimulates a particular cortical region, it sends indirect signals through connected neural pathways—much like how tapping the top of a drum can cause vibrations throughout its body. This allows stimulation of the cortex to ripple down and influence subcortical circuits, many of which are central to psychiatric and neurological function.
How TMS Reaches Deeper Brain Structures
How TMS Reaches Deeper Brain Structures
TMS primarily works by generating an electric field that induces action potentials in cortical neurons. These neurons, especially those in the prefrontal cortex, are directly connected to subcortical structures through well-established neural pathways. For example, the dorsolateral prefrontal cortex (DLPFC) is connected to the amygdala, which regulates emotional reactivity and fear. It also links with the hippocampus, involved in memory and stress regulation. TMS of motor areas, on the other hand, can influence the basal ganglia, which play a major role in movement and reward processing.
These circuits are bidirectional, meaning changes in cortical excitability can reshape subcortical activity—and vice versa. This is how TMS can modulate mood, motivation, emotional regulation, and cognitive performance, despite not directly stimulating those deeper areas.
Clinical Evidence of Subcortical Activation
Neuroimaging techniques such as fMRI, PET, and EEG have shown that TMS can induce changes in blood flow and activity patterns in deep brain areas. For instance:
- In major depressive disorder (MDD), studies have demonstrated that high-frequency TMS to the left DLPFC increases connectivity and normalizes activity in the subgenual anterior cingulate cortex (sgACC)—a key subcortical region implicated in mood regulation.
- In PTSD, low-frequency TMS to the right DLPFC has been found to reduce amygdala hyperactivity, contributing to a reduction in intrusive thoughts and emotional overdrive.
- In Parkinson’s disease, motor cortex stimulation appears to modulate the function of the basal ganglia and thalamus, helping to improve motor symptoms like rigidity and bradykinesia.
Importantly, these effects are not fleeting. Repeated stimulation over time has been shown to lead to lasting changes in connectivity and functional reorganization of both cortical and subcortical networks.
The Role of Subcortical Circuits in Mental Health
Subcortical regions are deeply involved in many conditions treated with TMS:
- The amygdala is central to anxiety, PTSD, and fear conditioning. Modulating its activity via TMS targeting the prefrontal cortex can help rebalance emotional responses.
- The hippocampus is linked to depression, especially when it comes to memory processing and rumination.
- The basal ganglia, often overactive in OCD, can be influenced by TMS through inhibitory protocols to reduce compulsive behavior.
- The thalamus, a relay station in the brain, is involved in sensory processing and consciousness and may be indirectly affected to stabilize mood and attention.
Understanding these relationships allows clinicians to select cortical targets strategically, knowing that their effects can propagate to deeper layers of the brain involved in the specific disorder.
The Promise of Deep TMS and Advanced Coils
While standard figure-eight coils are effective for modulating cortical networks, deep TMS (dTMS) expands the reach of stimulation to include subcortical areas more directly. These H-coils are engineered to penetrate deeper into the brain, delivering broader electric fields. dTMS has been FDA-approved for depression, OCD, and smoking cessation, and ongoing studies suggest it may also benefit conditions such as bipolar disorder and substance use disorders.
The ability of dTMS to stimulate deeper limbic structures like the insula and ACC opens new doors for treating emotional dysregulation and cognitive dysfunction. These structures are hard to reach using conventional coils but are increasingly recognized as critical hubs in mental health.
Looking Ahead
As technology advances, the precision of TMS is expected to improve through personalized mapping, functional imaging, and computational modeling. Future protocols may allow clinicians to select not just the surface target, but the downstream subcortical regions they wish to influence—enhancing both the specificity and impact of treatment.
In summary, TMS is not limited to surface-level effects. Its power lies in how it leverages the brain’s inherent connectivity, creating therapeutic change through cascades of neural communication that extend deep into subcortical terrain. This capacity helps explain why TMS can have such profound and enduring effects across a wide spectrum of mental and neurological disorders.
References
- Philip, N. S., Barredo, J., Aiken, E., & Carpenter, L. L. (2018). Neuroimaging mechanisms of therapeutic transcranial magnetic stimulation for major depressive disorder. Biological Psychiatry: Cognitive Neuroscience and Neuroimaging, 3(3), 211–222. https://doi.org/10.1016/j.bpsc.2017.10.007
- Fox, M. D., Buckner, R. L., White, M. P., Greicius, M. D., & Pascual-Leone, A. (2012). Efficacy of transcranial magnetic stimulation targets for depression is related to intrinsic functional connectivity with the subgenual cingulate. Biological Psychiatry, 72(7), 595–603. https://doi.org/10.1016/j.biopsych.2012.04.028
- Strafella, A. P., Paus, T., Barrett, J., & Dagher, A. (2001). Repetitive transcranial magnetic stimulation of the human prefrontal cortex induces dopamine release in the caudate nucleus. Journal of Neuroscience, 21(15), RC157. https://pubmed.ncbi.nlm.nih.gov/11459878/
- Zangen, A., Roth, Y., Voller, B., & Hallett, M. (2005). Transcranial magnetic stimulation of deep brain regions: Evidence for efficacy of the H-coil. Clinical Neurophysiology, 116(4), 775–779. https://pubmed.ncbi.nlm.nih.gov/15792886/



