How TMS Works on the Brain Part 8:Advances in Imaging for TMS Guidance
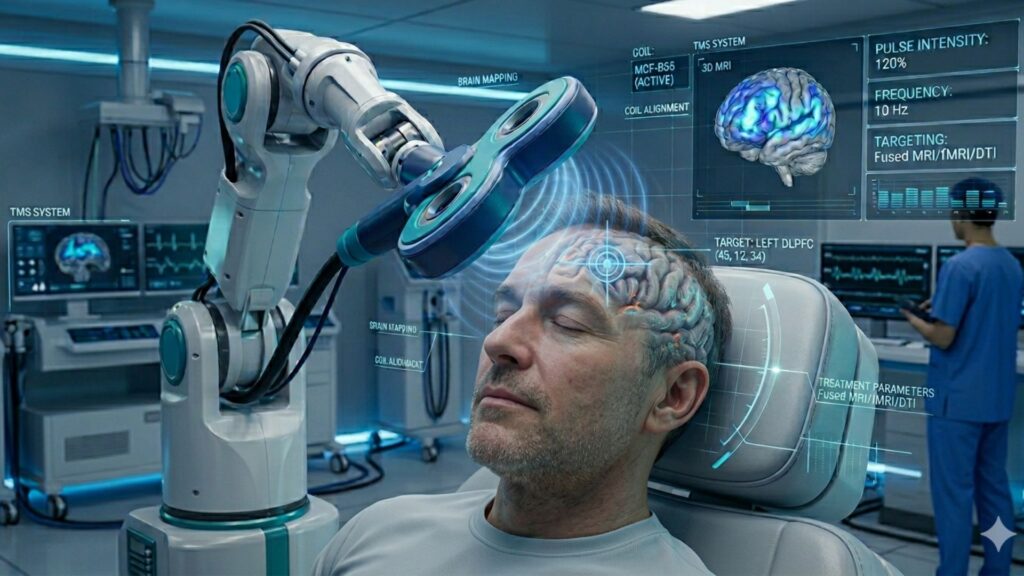
In recent years, one of the most exciting evolutions in transcranial magnetic stimulation (TMS) is the integration of advanced brain imaging. These technologies allow clinicians to move beyond “one-size-fits-all” treatment and toward highly personalized, precision-based care. Instead of relying solely on anatomical landmarks or generalized scalp coordinates, imaging techniques help identify exactly where to stimulate for the greatest therapeutic impact.
Why Imaging Matters in TMS
Traditional TMS protocols often use standard scalp-based measurements to locate treatment sites—like moving 5 to 6 centimeters anterior to the motor cortex to find the left dorsolateral prefrontal cortex (DLPFC). While this method can be effective, it doesn’t account for individual differences in brain anatomy or connectivity.
Modern imaging tools, however, provide clinicians with a real-time map of the patient’s brain. This helps pinpoint target regions more precisely, ensure consistent coil placement, and track how different areas respond over the course of treatment.
Types of Imaging Techniques Used
Several imaging technologies are now being used in TMS planning and monitoring:
- Magnetic Resonance Imaging (MRI): High-resolution structural MRI scans allow clinicians to visualize a patient’s unique brain anatomy. This is especially helpful for identifying the DLPFC, motor cortex, or more complex targets like the subgenual anterior cingulate cortex (sgACC).
- Functional MRI (fMRI): By tracking blood flow and activity levels, fMRI reveals which brain areas are underactive or hyperactive at rest or during specific tasks. This can guide clinicians toward areas needing stimulation.
- Electroencephalography (EEG) and Quantitative EEG (qEEG): EEG measures the brain’s electrical activity in real time. When combined with TMS, it provides feedback about how the brain is responding, enabling dynamic adjustments to protocols. qEEG takes this a step further by offering a statistical analysis of brainwave patterns, helping to identify atypical connectivity and guide personalized targeting. This is especially valuable in populations like autism, where individualized neural profiles vary widely.
- Diffusion Tensor Imaging (DTI): DTI tracks the integrity and direction of white matter tracts. This is useful for identifying how different regions are connected and predicting how stimulation in one area might influence others.
These tools support neuronavigation, which is a method for aligning the TMS coil with a specific brain target based on the patient’s own imaging data. Think of it like using GPS rather than a paper map—it adds accuracy, consistency, and safety.
Personalized Targeting and Improved Outcomes
The ability to personalize TMS based on imaging is revolutionizing treatment for conditions like depression, OCD, PTSD, and increasingly, autism spectrum disorder (ASD). For instance, researchers have found that stimulating areas functionally connected to the sgACC produces better outcomes in depression than stimulating based on surface landmarks alone.
In a 2021 study by Cash et al., patients who received TMS guided by their unique functional connectivity profiles saw significantly greater improvements than those treated with traditional coordinates. This shows that understanding how brain regions are wired together is just as important as knowing their location.
Personalized targeting is also being explored in emerging areas like autism, where imaging has shown that individuals with ASD often exhibit unique patterns of connectivity. These may include hypoconnectivity between the prefrontal cortex and social processing areas or hyperconnectivity in sensorimotor networks. Both fMRI and qEEG can be used to identify these irregularities. Early pilot studies are investigating how TMS can be used to normalize these imbalances and improve social cognition, executive function, and communication.
qEEG in particular offers a non-invasive and child-friendly way to assess brain function, making it especially valuable in pediatric or nonverbal populations with ASD. When paired with neuronavigated TMS, qEEG findings can help clinicians tailor stimulation protocols that address each individual’s specific brainwave anomalies. This approach shows promise in offering a non-invasive, individualized option for a population often underserved by traditional psychiatric treatments.
Additionally, real-time imaging feedback—especially using EEG—can help clinicians identify when a session is producing too much or too little activation, allowing for on-the-fly adjustments. This could improve safety, reduce side effects, and make each session more effective.
Looking Forward: AI and Real-Time Mapping
Emerging developments in artificial intelligence (AI) and machine learning are expected to further enhance imaging-guided TMS. Algorithms can already analyze imaging data to recommend optimal coil placement, intensity, and frequency patterns based on previous outcomes.
There is also growing interest in real-time functional mapping, where imaging or EEG is used during stimulation to monitor the brain’s response in real time. This could lead to adaptive protocols that evolve as the brain changes over the course of treatment—potentially accelerating results and reducing the number of sessions needed.
New technologies like connectome-based modeling and combined EEG-fMRI systems are also being explored for use in clinical settings, promising even greater personalization and precision in the near future. Connectome-based targeting, for instance, involves using whole-brain connectivity maps to identify ideal stimulation points that influence broader neural circuits. This approach has already shown promise in trials for depression and OCD and could be adapted for use in autism and neurodegenerative conditions.
Another promising technology is the use of wearable brain-monitoring devices that integrate EEG with TMS systems, providing portable, real-time brain feedback outside the clinic. These systems could make neuromodulation more accessible and continuous, bridging the gap between clinical sessions and everyday functioning.
Researchers are also investigating cloud-based platforms that analyze patient brain data remotely and feed insights back to clinicians, streamlining the workflow and improving consistency across treatment centers. These innovations not only enhance targeting but also support large-scale studies to refine TMS outcomes based on real-world data.
Conclusion
Advanced imaging has taken TMS from a promising therapy to a precision neuroscience tool. By using brain scans and connectivity maps to guide where and how stimulation occurs, clinicians can deliver more targeted, efficient, and effective treatment. As imaging continues to evolve and integrate with AI, the future of TMS looks not only brighter—but smarter.
References
- Cash, R. F. H., Cocchi, L., Lv, J., Wu, Y., Fitzgerald, P. B., & Zalesky, A. (2021). Personalized TMS targets based on individual functional connectivity for depression treatment. Biological Psychiatry Global Open Science, 1(2), 120–128. https://pubmed.ncbi.nlm.nih.gov/33544411/
- Fox, M. D., Buckner, R. L., White, M. P., Greicius, M. D., & Pascual-Leone, A. (2012). Efficacy of transcranial magnetic stimulation targets for depression is related to intrinsic functional connectivity with the subgenual cingulate. Biological Psychiatry, 72(7), 595–603. https://doi.org/10.1016/j.biopsych.2012.04.028
- Opitz, A., Zafar, N., Bockermann, V., Rohde, V., & Paulus, W. (2014). Validating computationally predicted TMS stimulation areas using MRI-guided neuronavigation. Brain Stimulation, 7(2), 234–242. https://pubmed.ncbi.nlm.nih.gov/24818076/
Conde, V., Tomasevic, L., Akopian, I., et al. (2019). The non-transcranial TMS-evoked potential is an inherent source of ambiguity in TMS–EEG studies. NeuroImage, 185, 300–312. https://www.sciencedirect.com/science/article/pii/S105381191832024X?via%3Dihub